Nomenclature
CAS number: 88-06-2
Dowicide 2S (Dow); Omal.
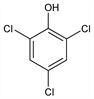
C
6H
3Cl
3O; mol wt 197.45.
C 36.50%, H 1.53%, Cl 53.87%, O 8.10%.
Description and references
Prepd by direct chlorination of phenol: Tiessens, Rec. Trav. Chim. 50, 115 (1931);
Chulkov et al., Org. Chem. Ind.
USSR 3, 97 (1937); Chem. Zentralbl. 1938, I, 1419; C.A. 31, 4967 (1937). Prepn of
sodium salt monohydrate: Hunter, Seyfried, J. Am. Chem. Soc. 43, 154 (1921). Review
of toxicology and human exposure: Toxicological
Profile for Chlorophenols (PB99-166639, 1999) 260 pp.
Properties
Crystals from ligroin. Strong phenolic odor.
Volatile with steam, but not from alkaline soln. d 1.4901. mp 69°. bp760 246°. Soly (g/100 g of solvent): Acetone
525; benzene 113; carbon tetrachloride 37; diacetone alcohol 335;
ether 354; denatured alcohol formula 30, 400; methanol 525; pine oil
163; Stoddard solvent 16; toluene 100; turpentine 37; water <0.1.Derivative
Sodium salt monohydrate.
Properties
Flaky crystals. Freely sol in water, alcohol,
ether, acetone. pH of satd aq soln 11.0-13.0.Caution
2,4,6-Trichlorophenol is reasonably
anticipated to be a human carcinogen: Report
on Carcinogens, Eleventh Edition (PB2005-104914, 2004)
p III-263.Use
Fungicide, bactericide, wood preservative, biocide;
intermediate in production of higher chlorinated phenols.
