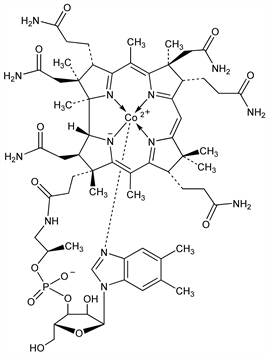
10016. Cob(II)alamin
Nomenclature
Description and references
A one-electron reduction product of vitamin B12 containing a 5-coordinate 2+ cobalt atom. Undergoes a further one-electron reduction to cob(I)alamin. Intermediate in biosynthesis of cobamamide and in cobalamin-dependent reactions. Prepn: H. Diehl, R. Murie, Iowa State Coll. J. Sci. 26, 555 (1952); R. O. Brady, H. A. Barker, Biochem. Biophys. Res. Commun. 4, 373 (1961) DOI; H.-U. Blaser, J. Halpern, J. Am. Chem. Soc. 102, 1684 (1980) DOI. Valence of reduced forms of B12: H. A. O. Hill et al., Chem. Ind. (London) 1964, 197. Crystal structure: B. Krutler et al., J. Am. Chem. Soc. 111, 8936 (1989) DOI; of biosynthetic intermediates: M. St. Maurice et al., Biochemistry 47, 5755 (2008) DOI PubMed. Review of prepns: D. Dolphin, Methods Enzymol. 18, (Pt. C), 34-52 (1971) DOI; of role in enzymatic reactions: J. M. Pratt, Pure Appl. Chem. 65, 1513 (1993) DOI; G. H. Reed, S. O. Mansoorabadi, Curr. Opin. Struct. Biol. 13, 716-721 (2003) DOI PubMed; M. Yamanishi et al., Trends Biochem. Sci. 30, 304-308 (2005) DOI PubMed.