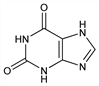
10059. Xanthine
Nomenclature
CAS number: 69-89-6
3,7-Dihydro-1H-purine-2,6-dione; 2,6(1H,3H)-purinedione; 2,6-dioxopurine. C5H4N4O2; mol wt 152.11.
C 39.48%, H 2.65%, N 36.83%, O 21.04%.
Description and references
Occurs in animal organs, yeast, potatoes, coffee beans, tea. First isolated from urinary bladder stones: Beilstein 26, 447 (1937). Prepd by treating a sulfuric acid soln of guanine with sodium nitrite: Fischer, Ann. 215, 309 (1882). Several other syntheses, cf. Levene, Bass, Nucleic Acids (New York, 1931).