10169. Zinquin
Nomenclature
CAS number: 151606-29-0
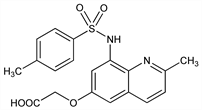
[[2-Methyl-8-[[(4-methylphenyl)sulfonyl]amino]-6-quinolinyl]oxy]acetic
acid; Zinquin A. C19H18N2O5S; mol wt 386.42.
C 59.06%, H 4.70%, N 7.25%, O 20.70%, S 8.30%.
Description and references
TSQ, q.v., derived membrane permeant fluorescent probe selective for Zn2+ in the presence of Ca2+ and Mg2+. Synthesis: I. B. Mahadevan et al., Aust. J. Chem. 49, 561 (1996). Coordination chemistry: C. J. Fahrni, T. V. O'Halloran, J. Am. Chem. Soc. 121, 11448 (1999); potentiometric coordination of fluorophore: K. M. Hendrickson et al., ibid. 125, 3889 (2003). Visualization of intracellular Zn2+ pools in pancreatic islets: P. D. Zalewski et al., J. Histochem. Cytochem. 42, 877 (1994); in apoptosis: P. J. Smith et al., Am. J. Physiol. Cell Physiol. 283, C609 (2002); G. R. Sauer et al., J. Cell. Biochem. 88, 954 (2003).
Properties
Crystals from ethyl acetate or ethyl acetate/light petroleum, mp 198-200°. pK1 (quinoline nitrogen): <2; pK (carboxylic acid proton): 5.77 ±0.03; pK (sulfonamide proton): 9.91±0.02.Derivative
Ethyl Ester.Nomenclature
CAS number: 181530-09-6
Zinquin E. C21H22N2O5S; mol wt 414.47.
C 60.85%, H 5.35%, N 6.76%, O 19.30%, S 7.74%.