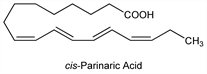
10237. Parinaric Acid
Nomenclature
Description and references
Close structural analog of intrinsic membrane lipids. The naturally occuring cis-form is found in the seeds of the tropical rainforest Makita tree, indigenous to Fiji. Isoln from the kernel oil of Parinarium laurinum A. Gray (Rosaceae): M. Tsujimoto, H. Koyanagi, Kogyo Kagaku Zasshi 36, 110 (1933). Stereochemical synthesis of isomers: H. P. Kaufmann, R. K. Sud, Ber. 92, 2797 (1959) DOI; D. V. Kuklev, W. L. Smith, Chem. Phys. Lipids 131, 215 (2004) DOI PubMed. Identification of α-parinaric acid as the 9Z,11E,13E,15Z-isomer: F. D. Gunstone, R. Subbarao, ibid. 1, 349 (1967) DOI. Characterization as a fluorescent probe for phospholipid membrane studies: L. A. Sklar et al., Proc. Natl. Acad. Sci. USA 72, 1649 (1975) DOI PubMed; idem et al., Biochemistry 16, 813, 819, 829 (1977) DOI PubMed DOI PubMed DOI PubMed. Review of lipid partition behavior: idem, Mol. Cell. Biochem. 32, 169-177 (1980) DOI PubMed. Use as a fluorescent probe for lipid peroxidation: F. A. Kuypers et al., Biochim. Biophys. Acta 921, 266 (1987) PubMed; for lipase activity: A. M. Rogel et al., Lipids 24, 518 (1989) DOI PubMed.