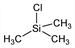10311. Chlorotrimethylsilane
Nomenclature
Description and references
Silylating reagent and Lewis acid catalyst in synthetic organic chemistry. Prepn from trimethylsilane and chlorine: A. G. Taylor, B. V. De G. Walden, J. Am. Chem. Soc. 66, 842 (1944) DOI; from silicon chloride: H. S. Booth, J. F. Suttle, ibid. 68, 2658 (1946) DOI; from hexamethyldisiloxane and ammonium chloride: B. O. Pray et al., ibid. 70, 433 (1948) DOI. Crystal structure: J. Buschmann et al., Acta Crystallogr. C56, 121 (2000) DOI. Thermodynamic studies: A. E. Beezer, C. T. Mortimer, J. Chem. Soc. A 1966, 514 DOI. Synthetic applications: G. A. Olah et al., J. Org. Chem. 44, 4272 (1979) DOI; J. G. Lee et al., Tetrahedron Lett. 31, 6677 (1990) DOI; J.-M. Lin, B.-S. Liu, Synth. Commun. 27, 739 (1997) DOI; J. Eras et al., J. Org. Chem. 67, 8631 (2002) DOI PubMed; in catalysis: P. Verma, S. Ray, Indian J. Chem. 29B, 652 (1990); L.-W. Xu et al., Synth. Commun. 37, 3095 (2007) DOI. Derivitization and gas chromatography: J. Eras et al., J. Chromatogr. A 1047, 157 (2004) DOI PubMed.