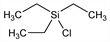
10397. Chlorotriethylsilane
Nomenclature
Description and references
Silylating reagent used in organic synthesis to introduce the triethylsilyl (TES) protecting group. Prepn from triethylethoxysilane: A. Ladenburg, Ann. 164, 300 (1872); from hexaethyldisiloxane: P. A. Di Giorgio et al., J. Am. Chem. Soc. 68, 1380 (1946) DOI; from β-chloroethyltriethylsilane: L. H. Sommer, ibid. 70, 2869 (1948) DOI. Use in protection of hydroxyl groups: W. Oppolzer et al., Helv. Chim. Acta 64, 2002 (1981) DOI; W. R. Roush, S. Russo-Rodriguez, J. Org. Chem. 52, 598 (1987) DOI. Additional synthetic applications: S. Danishefsky et al., J. Am. Chem. Soc. 107, 1285 (1985) DOI; Y. Fujii et al., J. Organomet. Chem. 692, 375 (2007) DOI. Review: E. Turos in Encyclopedia of Reagents for Organic Synthesis 2, L. A. Paquette, Ed. (Wiley, New York, 1995) pp 1225-1227.