10322. Eribulin
Nomenclature
CAS number: 253128-41-5
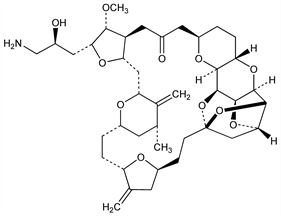
(2R,3R,3aS,7R,8aS,9S,10aR,11S,12R,13aR,13bS,15S,18S,21S,24S,26R,28R,29aS)-2-[(2S)-3-Amino-2-hydroxypropyl]hexacosahydro-3-methoxy-26-methyl-20,27-bis(methylene)-11,15:18,21:24,28-triepoxy-7,9-ethano-12,15-methano-9H,15H-furo[3,2-i]furo[2′,3′:5,6]pyrano[4,3-b][1,4]dioxacyclopentacosin-5(4H)-one; ER-086526. C40H59NO11; mol wt 729.90.
C 65.82%, H 8.15%, N 1.92%, O 24.11%.
Description and references
Synthetic analog of the marine polyether macrolide, halichondrin B. Inhibits tumor cell proliferation by suppressing microtubule growth. Prepn: B. Littlefield et al., WO 9965894 (1999); eidem, US 6214865 (2001 to Eisai); W. Zheng et al., Bioorg. Med. Chem. Lett. 14, 5551 (2004) DOI PubMed. Anticancer activities: M. J. Towle et al., Cancer Res. 61, 1013 (2001) PubMed. Mechanism of action: M. A. Jordan et al., Mol. Cancer Ther. 4, 1086 (2005) DOI PubMed. Review of pharmacology and clinical experience: S. Newman, Curr. Opin. Investig. Drugs 8, 1057-1066 (2007) PubMed.
Derivative
Monomethanesulfonate.Nomenclature
CAS number: 441045-17-6
Eribulin mesylate; E-7389. C40H59NO11.CH3SO3H; mol wt 826.00.
C 59.62%, H 7.69%, N 1.70%, O 27.12%, S 3.88%.
Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Antibiotics and Analogs