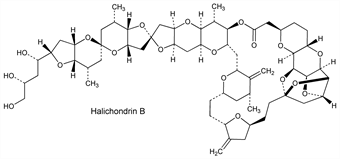
10323. Halichondrins
Description and references
Group of antimitotic, polyether macrolides produced by several marine sponges. Halichondrin B has the most potent bioactivity and is the prototype for the anticancer agent, eribulin, q.v. Isoln from Halichondria okadai Kadota: D. Uemura et al., J. Am. Chem. Soc. 107, 4796 (1985) DOI; and antitumor activity: Y. Hirata, D. Uemura, Pure Appl. Chem. 58, 701 (1986) DOI. Isoln from Axinella sp.: G. R. Pettit et al., J. Med. Chem. 34, 3339 (1991) DOI PubMed. Total synthesis of halichondrin B and norhalichondrin B: T. D. Aicher et al., J. Am. Chem. Soc. 114, 3162 (1992) DOI. Effect on bovine brain tubulin: R. F. Luduena et al., Biochem. Pharmacol. 45, 421 (1993) DOI PubMed. Review of potential production methods: D. Sipkema et al., Biotechnol. Bioeng. 90, 201-222 (2005) DOI PubMed.