12-03682. Erdin [Archived]
Nomenclature
CAS number: 26891-81-6
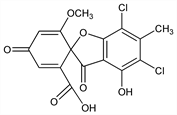
5,7-Dichloro-4-hydroxy-6′-methoxy-6-methyl-3,4′-dioxospiro[benzofuran-2(3H),1′-[2,5]cyclohexadiene]-2′-carboxylic acid; dl-erdin. C16H10Cl2O7; mol wt 385.15.
C 49.90%, H 2.62%, Cl 18.41%, O 29.08%.
Description and references
Chlorine containing fungal metabolite isolated from Aspergillus terreus Thom, as the racemate, together with geodin: H. Raistrick, G. Smith, Biochem. J. 30, 1315 (1936). Characterization and structure of erdin and geodin: P. W. Clutterbuck et al., ibid. 31, 1089 (1937); C. T. Calam et al., ibid. 33, 579 (1939); eidem, ibid. 41, 458 (1947); D. H. Barton, A. I. Scott, J. Chem. Soc. 1958, 1767. Biosynthesis of geodin: Rhodes et al., Chem. Ind. (London) 1962, 611.
Properties
Needles, mp 210-212°. uv max: 284 nm (ε 21000).Derivative
(+)-Form.Nomenclature
CAS number: 3405-51-4
Properties
Prisms from chloroform + light petr, mp 210-212°. [α]D +149° (c = 0.41 in dioxane). uv max: 284 nm (ε 21000). Reacts as dibasic acid, dec slowly in presence of alkali. Solubilities of erdin are closely similar to those of geodin except that it is only very slightly sol in chloroform.Derivative
(+)-Form methyl ester.Nomenclature
CAS number: 427-63-4
Geodin. C17H12Cl2O7; mol wt 399.18.
C 51.15%, H 3.03%, Cl 17.76%, O 28.06%.
Properties
Prisms from chloroform + ether, mp 228-231°. [α]D +140° (c = 0.8). uv max: 284 nm (ε 19000). Practically insol in water, petr ether. Very sparingly sol in benzene, ether; somewhat more sol in alc, ethyl acetate; readily sol in chloroform, acetone, dioxane.Status
This monograph has been retired and is no longer subject to revision or update.