12-08984. Streptomycin B [Archived]
Nomenclature
CAS number: 128-45-0
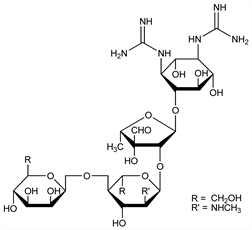
Mannosidostreptomycin; mannosylstreptomycin. C27H49N7O17; mol wt 743.71.
C 43.60%, H 6.64%, N 13.18%, O 36.57%.
Description and references
Isoln from streptomycin concentrates: Fried, Titus, J. Biol. Chem. 168, 391 (1947); 174, 57 (1948); Fried, Stavely, J. Am. Chem. Soc. 69, 1549 (1947); Fried, Titus, ibid. 70, 3615 (1948); O'Keeffe et al., ibid. 71, 2452 (1949); see also Langlykke, Perlman, US 2493489 (1950 to Squibb). Structure: Fried, Stavely, 113th Meeting Am. Chem. Soc., Chicago, Ill. (April 20, 1948) Abstracts of Papers p 25C; Peck et al., J. Am. Chem. Soc. 70, 3968 (1948); Stavely, Fried, ibid. 71, 135 (1949); 74, 5461 (1952).
Properties
On catalytic hydrogenation absorbs approx one mole of hydrogen and forms dihydrostreptomycin B. Yields maltol on alkaline hydrolysis. When treated with dil acids liberates streptidine (or an isomer of it) isolated as the sulfate monohydrate, C8H18N6O4.H2SO4.H2O.Derivative
Trihydrochloride monohydrate.C27H49N7O17.3HCl.H2O; mol wt 871.11.
C 37.23%, H 6.25%, N 11.26%, O 33.06%, Cl 12.21%.
Properties
White, amorphous powder; mp 190-200° (corr, decompn) after drying in vacuo at 100°. [α]D25 47° (c = 1.35 in water). Appears to have about one-fourth of the antibiotic activity of streptomycin trihydrochloride.Status
This monograph has been retired and is no longer subject to revision or update.