13-08904. Streptolydigin [Archived]
Nomenclature
CAS number: 7229-50-7
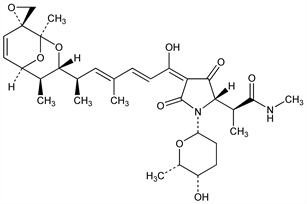
(αS,2S,4E)-4-[(2E,4E,6R)-6-[(1R,2′R,3R,4S,5R)-1,4-Dimethylspiro[2,9-dioxabicyclo[3.3.1]non-6-ene-8,2′-oxiran]-3-yl]-1-hydroxy-4-methyl-2,4-heptadienylidene]-N,α-dimethyl-3,5-dioxo-1-[(2S,5S,6S)-tetrahydro-5-hydroxy-6-methyl-2H-pyran-2-yl]-2-pyrrolidineacetamide; Portamycin (Upjohn). C32H44N2O9; mol wt 600.70.
C 63.98%, H 7.38%, N 4.66%, O 23.97%.
Description and references
Antibiotic isolated from culture filtrates of Streptomyces lydicus n. sp.: Eble et al., Antibiot. Annu. 1955-1956, 893; GB 779570 (1957 to Upjohn). Structure: Rinehart et al., J. Am. Chem. Soc. 85, 4083 (1963). Stereochemistry: Duchamp et al., ibid. 95, 4077 (1973). RNA-polymerase inhibitor: Cassani et al., Nature New Biol. 230, 197 (1971). NMR studies: V. J. Lee, K. L. Rinehart, J. Antibiot. 34, 408 (1980). Toxicity study: DeBoer et al., Antibiot. Annu. 1955-1956, 886.
Properties
Crystals from acetone + water, mp 147-148°. [α]D25 93° (c = 1.6 in chloroform). uv max (0.01N ethanolic KOH): 261, 291, 336 nm (E1%1cm 223.6, 270.7, 331.0); uv max (0.01N ethanolic H2SO4): 234, 357.5, 370 nm (E1%1cm 139.1, 590.5, 560.3). Sol in ethanol, ethyl acetate, ether, chloroform. Practically insol in water, hydrocarbons. LD50 i.p. in mice: 533 mg/kg (DeBoer).Derivative
Sodium salt.Nomenclature
CAS number: 80081-26-1
C32H43N2NaO9; mol wt 622.68.
C 61.72%, H 6.96%, N 4.50%, Na 3.69%, O 23.12%.
Properties
mp 225°. [α]D25 +153° (c = 1.35 in chloroform). Sol in the usual organic solvents and in water. Practically insol in hydrocarbons.Therapeutic Category
Antibacterial.
Status
This monograph has been retired and is no longer subject to revision or update.