13-01838. β-Carboxyaspartic Acid [Archived]
Nomenclature
Description and references
Amino acid found in ribosomal proteins of E. coli; homolog of γ-carboxyglutamic acid. Isoln and synthesis of dl-form: M. R. Christy et al., J. Am. Chem. Soc. 103, 3935 (1981). Synthesis of dl-form: E. B. Henson et al., Tetrahedron 37, 2561 (1981); N. E. Dixon, A. M. Sargeson, J. Am. Chem. Soc. 104, 6716 (1982). Quantitative analysis method: P. V. Hauschka et al., Anal. Biochem. 108, 57 (1980). Chemical reactivity: M. R. Christy, T. H. Koch, J. Am. Chem. Soc. 104, 1771 (1982). Crystal structure and pKa values of dl-zwitterion: B. Richey et al., Biochemistry 21, 4819 (1982). Asymmetric synthesis of d-form: R. M. Williams et al., J. Am. Chem. Soc. 110, 482 (1988). Review: T. H. Koch et al., Methods Enzymol. 107, 563-575 (1984).
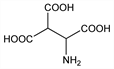
Derivative
dl-Form hydrochloride.Nomenclature
Properties
White crystalline powder. pK1 0.8 ±0.2, pK2 2.5 ±0.1, pK3 4.7 ±0.1, pK4 10.9 ±0.1.Derivative
d-Form hydrochloride.Nomenclature
Properties
White solid, [α]D25 13.25° (c = 0.8 in H2O, pH 2.56).Status
This monograph has been retired and is no longer subject to revision or update.