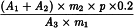Valerian Dry Hydroalcoholic Extract
Ph Eur
DEFINITION
Extract produced from Valerian root (0453).
Content
Minimum 0.25 per cent m/m of sesquiterpenic acids, expressed as valerenic acid (C15H22O2; Mr 234.3) (anhydrous extract).
PRODUCTION
The extract is produced from the herbal drug by a suitable procedure using ethanol (30-90 per cent V/V) or methanol (40-55 per cent V/V).
CHARACTERS
Appearance
Brown, hygroscopic powder.
IDENTIFICATION
Thin-layer chromatography (2.2.27).
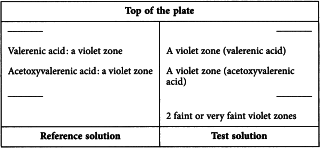
Test solution Suspend 1 g of the extract to be examined in 10 mL of methanol R and sonicate for 10 min. Filter the supernatant through a membrane filter (nominal pore size 0.45 µm).
Reference solution Dissolve 5 mg of acetoxyvalerenic acid R and 5 mg of valerenic acid R in 20 mL of methanol R.
PlateTLC silica gel plate R (5-40 µm) [or TLC silica gel plate R (2-10 µm)].
Mobile phaseglacial acetic acid R, ethyl acetate R, cyclohexane R (2:38:60 V/V/V).
Application 20 µL [or 5 µL] as bands of 10 mm [or 8 mm].
Development Over a path of 10 cm [or 6 cm].
Drying In air.
Detection Treat with anisaldehyde solution R and heat at 100-105 °C for 5-10 min; examine in daylight.
Results See below the sequence of zones present in the chromatograms obtained with the reference solution and the test solution. Furthermore, other violet zones may be present in the chromatogram obtained with the test solution.
TESTS
Water (2.5.12)
Maximum 5.0 per cent, determined on 0.5 g.
ASSAY
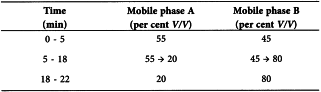
Liquid chromatography (2.2.29).
Test solution Suspend 1.00 g of the extract to be examined in 50.0 mL of methanol R1, sonicate for 10 min and filter through a membrane filter (nominal pore size 0.45 µm).
Reference solution Dissolve a quantity of valerian dry extract HRS corresponding to 0.5 mg of valerenic acid in methanol R1 and dilute to 10.0 mL with the same solvent. Sonicate for 10 min and filter through a membrane filter (nominal pore size 0.45 µm).
Flow rate 1.5 mL/min.
Detection Spectrophotometer at 220 nm.
Injection 20 µL.
Identification of peaks Use the chromatogram supplied with valerian dry extract HRS and the chromatogram obtained with the reference solution to identify the peaks due to hydroxyvalerenic acid, acetoxyvalerenic acid and valerenic acid.
Calculate the percentage content of sesquiterpenic acids, expressed as valerenic acid, using the following expression:
A1 | = | area of the peak due to hydroxyvalerenic acid in the chromatogram obtained with the test solution; |
A2 | = | area of the peak due to acetoxyvalerenic acid in the chromatogram obtained with the test solution; |
A3 | = | area of the peak due to valerenic acid in the chromatogram obtained with the test solution; |
A4 | = | area of the peak due to valerenic acid in the chromatogram obtained with the reference solution; |
m1 | = | mass of the extract to be examined used to prepare the test solution, in grams; |
m2 | = | mass of valerian dry extract HRS used to prepare the reference solution, in grams; |
p | = | percentage content of valerenic acid in valerian dry extract HRS. |
Ph Eur