|
|
|
|
|
|
|
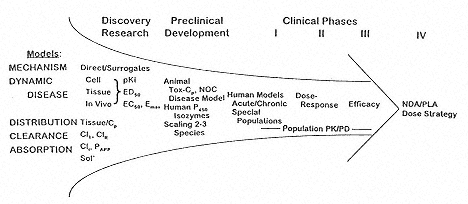
Managing the type of information collected and knowing how it will be used should be a critical advantage in drug development. Figure 9 diagrams a macro-perspective for collecting PK/PD information from discovery research to NDA filing. This integrated drug development concept was recommended by Peck et al. [18]. Creating this type of framework allows the scientist to understand the relationship of their contribution to the whole process. This orderly supply of information will feed the project team and prompt them to make decisions. The models provide an effective means for the skill areas to infuse wisdom into the project team. |
|
|
|
|
|
|
|
|
Managing and organizing the information flow is critical to making rapid development decisions in early clinical development. In a sense, the most important value for clinical pharmacology information is in making rapid, effective drug development decisions within the company either in the form of a go/no go decision or in designing subsequent studies. While the studies tend to be small in patient numbers, there are many protocols that usually do not take more than weeks to complete the clinical portion. Using remote data entry for all clinical data and collecting analytical information closer to sample collection from the patient should allow development decisions to be made more rapidly and study reports to be issued within weeks of the last patient studied as opposed to months or years. A close partnership between the clinical pharmacology clinical monitor, data management, and information technology staffs is required to accomplish this goal. |
|
|
|
|
|
|
|
|
 |
|
|
|
|
|
|
|
|
Fig. 9
PK/PD information flow paradigm from discovery research to the NDA/PLA
dosing strategy. |
|
|
|
|
|