260. Allethrins
Nomenclature
Allyl cinerins.Description and references
Synthetic analogues of the naturally occurring insecticides cinerins, jasmolins, and pyrethrins, q.q.v. Review: Barthel, World Rev. Pest Control 6, 59 (1967); World Health Org., Environ. Health Criter. 87, 1-75 (1989). Review of toxicology and human exposure: Toxicological Profile for Pyrethrins and Pyrethroids (PB2004-100004, 2003) 332 pp.
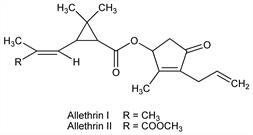
Derivative
Allethrin I.Nomenclature
Description and references
Synthesis: M. S. Schechter et al., J. Am. Chem. Soc. 71, 1517, 3165 (1949); H. A. Stansbury, H. R. Guest, US 2768965 (1956 to UCC). Commercial process: H. J. Sanders, A. W. Taff, Ind. Eng. Chem. 46, 414 (1954).
Properties
The commercial product is a mixture of 8 optically active isomers. Clear, pale yellow oil. Poisonous. bp 0.1 140°. Decomp by rapid pyrolysis at >400°. Vaporizes without dec when heated at 150°. Unstable in light, air, alkaline conditions. d2020 1.010; nD20 1.5040; nD30 1.5023. Practically insol in water. Sol in alcohol, petr ether, kerosene, carbon tetrachloride, ethylene dichloride, nitromethane. Incompatible with alkalies.Derivative
Allethrin II.Nomenclature
Description and references
Synthesis: M. Matsui, Y. Yamada, Agric. Biol. Chem. 27, 373 (1963); M. Matsui, H. Meguro, ibid. 28, 27 (1964); M. Elliott, N. F. Janes, Chem. Ind. (London) 1969, 270; A. Kobayashi et al., Agric. Biol. Chem. 35, 1961 (1971); T. Sugiyama et al., ibid. 36, 565 (1972).