2396. Clopidogrel
Nomenclature
Description and references
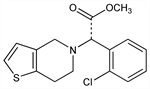
Analog of ticlopidine, q.v.; inhibits ADP-induced platelet aggregation. Prepn (unspec. stereochem.): D. Aubert et al., EP 99802; eidem, US 4529596 (1984, 1985 both to Sanofi). Prepn of (+)-form: A. Badorc, D. Fréhel, EP 281459; eidem, US 4847265 (1988, 1989 both to Sanofi). Clinical pharmacology and mechanism of action: D. C. B. Mills et al., Arterioscler. Thromb. 12, 430 (1992). Clinical trial vs aspirin for prevention of atherothrombotic events: CAPRIE Steering Committee, Lancet 348, 1329 (1996). Review of pharmacology and therapeutic potential: G. Feuerstein et al., Expert Opin. Invest. Drugs 4, 425-430 (1995); of use in secondary prevention in stroke: H.-C. Diener et al., Expert Opin. Pharmacother. 6, 755-764 (2005).
Properties
Colorless oil. [α]D20 +51.52° (c = 1.61 in methanol).Derivative
Hydrogen sulfate.Nomenclature
Properties
White crystals, mp 184°. [α]D20 +55.10° (c = 1.891 in methanol). Sol in water, methanol.Therapeutic Category
Antithrombotic.
Keywords
Antithrombotic