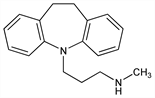
2920. Desipramine
Nomenclature
Description and references
Prepn: GB 908788; BE 614616 (both 1962 to Geigy); J. H. Biel, C. I. Judd, GB 980231 (1965 to Lakeside); eidem, US 3454554 (1969 to Colgate Palmolive). Chemistry, pharmacology: E. Eriksoo, O. Rohte, Arzneim.-Forsch. 20, 1561 (1970). Clinical response, plasma levels, pharmacokinetics: P. D. Hrdina, Y. D. Lapierre, Prog. Neuro-Psychopharmacol. 4, 591 (1980). Mechanism of action study: S. A. Checkley et al., Br. J. Psychiatry 138, 248 (1981). Efficacy in depression: W. Z. Potter et al., Psychopharmacol. Bull. 17, 26 (1981); J. W. Stewart et al., ibid. 136. Teratological study: L. Aeppli, Arzneim.-Forsch. 19, 1617 (1969). Carcinogenicity and mutagenicity study: H. Kubinski et al., Mutat. Res. 89, 95 (1981). Has been found to be one of the most effective cmpds described for in vitro and in vivo reversal of chloroquine, q.v., resistance in Plasmodium falciparum: A. J. Bitonti et al., Science 242, 1301 (1988).
Properties
bp0.02 172-174°. uv max: 213, 252 nm (log ε 4.39, 3.93).Derivative
Hydrochloride.Nomenclature
Properties
Crystals from methanol ether, mp 215-216°. Sol in water. LD50 in mice, rats (mg/kg): 500, 385 orally; 94, 48 i.p.; 420, 183 s.c. (Eriksoo, Rohte).Therapeutic Category
Antidepressant.
Keywords
Antidepressant; Tricyclics