5586. Lovastatin
Nomenclature
Description and references
Fungal metabolite; potent inhibitor of HMG-CoA reductase, the rate controlling enzyme in cholesterol biosynthesis. Isoln from Monascus ruber: A. Endo, J. Antibiot. 32, 852 (1979); from Aspergillus terreus: R. L. Monaghan et al., US 4231938 (1980 to Merck & Co.). Structure and biochemical properties: A. W. Alberts et al., Proc. Natl. Acad. Sci. USA 77, 3957 (1980). Total synthesis: M. Hirama, M. Iwashita, Tetrahedron Lett. 24, 1811 (1983). Review of syntheses: T. Rosen, C. H. Heathcock, Tetrahedron 42, 4909-4951 (1986). Biosynthesis: M. D. Greenspan, J. B. Yudkovitz, J. Bacteriol. 162, 704 (1985); R. N. Moore et al., J. Am. Chem. Soc. 107, 3694 (1985). HPLC determn in plasma and bile: R. J. Stubbs et al., J. Chromatogr. 383, 438 (1986). Clinical pharmacology: S. M. Grundy, G. L. Vega, J. Lipid Res. 26, 1464 (1985). Clinical comparison with gemfibrozil, q.v.: M. J. Tikkanen et al., Am. J. Cardiol. 62, 35J (1988). Review of clinical experience: J. A. Tobert, Am. J. Cardiol. 62, 28J-34J (1988). Comprehensive description: G. S. Brenner et al., Anal. Profiles Drug Subs. Excip. 21, 277-305 (1992). Prevention of acute coronary events in men and women with average cholesterol levels: J. R. Downs et al., J. Am. Med. Assoc. 279, 1615 (1998). Review of development and clinical experience with extended-release niacin, q.v.: H. E. Bays, Expert Rev. Cardiovasc. Ther. 2, 485-501 (2004). Review of therapeutic efficacy in high risk populations: A. Frisinghelli, A. Mafrici, Clin. Drug Invest. 27, 591-604 (2007).
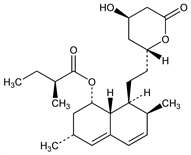
Properties
White crystals, mp (under N2): 174.5°. [α]D25 +323° (c = 0.5 g in 100 ml acetonitrile). uv max: 231, 238, 247 nm (A1% 532, 621, 418). Soly at room temp (mg/ml): acetone 47, acetonitrile 28, n-butanol 7, i-butanol 14, chloroform 350, N,N-dimethylformamide 90, ethanol 16, methanol 28, n-octanol 2, n-propanol 11, i-propanol 20, water 0.4 × 103. LD50 orally in mice: >1000 mg/kg (Endo).Therapeutic Category
Antilipemic.
Keywords
Antilipemic; HMG CoA Reductase Inhibitors; HMG CoA Reductase Inhibitor