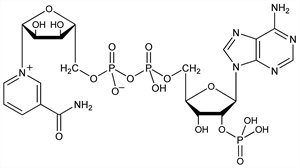
6348. NADP
Nomenclature
Description and references
One of the biologically active forms of nicotinic acid, q.v. Differs from NAD, q.v., by an additional phosphate group at the 2′-position of the adenosine moiety. Serves as a coenzyme of hydrogenases and dehydrogenases. Present in living cells primarily in the reduced form (NADPH) and is involved in synthetic reactions. Isoln from horse blood: O. Warburg et al., Biochem. Z. 282, 157 (1935); from hog liver by adsorption on charcoal: G. A. LePage, G. C. Mueller, J. Biol. Chem. 180, 975 (1949); from sheep liver by extraction with hot water: A. Kornberg, B. L. Horecker, Biochem. Prep. 3, 24 (1953); from human red blood cells by anion-exchange chromatography: G. R. Bartlett, J. Biol. Chem. 234, 449, 459 (1959). Chemical and enzymatic synthesis: D. R. Walt et al., J. Am. Chem. Soc. 106, 234 (1984). Nomenclature: M. Dixon, Science 132, 1548 (1960). Review of synthesis and metabolism: Y. Nishizuka in Method. Chim. vol. 11, F. Korte, Ed. (Academic Press, New York, 1977) pp 84-87. Review: “Niacin: Nicotinic Acid, Nicotinamide, NAD(P)” in Vitamins, W. Friedrich, Ed. (Walter de Gruyter, Berlin, 1988) pp 473-542.