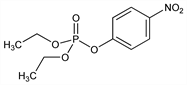
7030. Paraoxon
Nomenclature
Description and references
Prepd by the action of diethyl chlorophosphate on sodium p-nitrophenolate or by nitration of diethyl phenyl phosphate: Schrader, BIOS Final Report No. 714, 52 (1947); Angew. Chem. 62, 471 (1950); CH 257649 (1949); Fagerlind et al., Sven. Farm. Tidskr. 56, 303, C.A. 46, 9259. Physical properties: E. F. Williams, Ind. Eng. Chem. 43, 950 (1951). Formation from parathion on citrus foliage and soil surface: R. C. Spear et al., J. Agric. Food Chem. 23, 808 (1975); W. F. Spencer et al., Bull. Environ. Contam. Toxicol. 14, 265 (1975). Toxicity study: W. R. Pickering, J. C. Malone, Biochem. Pharmacol. 16, 1183 (1967). Brief review: G. Schrader, Die Entwicklung neuer insektizider Phosphors"aure-Ester (Verlag Chemie, Weinheim, 1963) pp 259-272.