8330. Salicylanilide
Nomenclature
CAS number: 87-17-2
2-Hydroxy-N-phenylbenzamide; N-phenylsalicylamide; Salinidol (Doak). C13H11NO2; mol wt 213.23.
C 73.23%, H 5.20%, N 6.57%, O 15.01%.
Description and references
Usually made by the reaction of salicylic acid with aniline in the presence of PCl3 at an elevated temp. Better yields are obtained by using an inert organic solvent such as toluene or carbon tetrachloride as a reaction diluent. Novel process using ion-exchange resins: Majewski, Skelly, US 3221051 (1965). Alternate process: Majewski et al., US 3231611 (1966 to Dow).
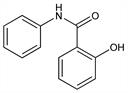
Properties
Odorless leaflets. mp 135.8-136.2°. Slightly sol in water; freely sol in alcohol, ether, chloroform, benzene.Caution
In concd form may cause irritation of skin, mucous membranes. See also Salicylic Acid.Use
Anti-mildew, fungicide.Therapeutic Category
Antifungal (topical).
Therapeutic Category (Veterinary)
Antifungal (topical).Keywords
Antifungal (Synthetic)