Nomenclature
CAS number: 1746-01-6
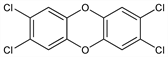
2,3,7,8-Tetrachlorodibenzo[
b,e][1,4]dioxin; 2,3,7,8-tetrachlorodibenzo-
p-dioxin; 2,3,6,7-tetrachlorodibenzodioxin; dioxin; TCDBD.
C
12H
4Cl
4O
2; mol wt 321.97.
C 44.76%, H 1.25%, Cl 44.05%, O 9.94%.
Description and references
Highly toxic contaminant; produced as a by-product
during the manuf of chlorinated phenols (2,4,5-trichlorophenol, q.v.) and phenoxyherbicides (2,4-D and 2,4,5-T, q.q.v.),
chlorine bleaching of paper pulp and combustion of chlorine-containing
waste. Prepn: W. Sandermann, Ber. 90, 690 (1957); M. Tomita et al., Yakugaku Zasshi 79, 186 (1959), C.A. 53, 13152d (1959). Crystal
structure: F. P. Boer et al., Acta Crystallogr. 28B, 1023 (1972). Environmental
degradation: D. G. Crosby, A. S. Wong, Science 195, 1337 (1976). Comprehensive review of formation,
chemistry, and toxic and environmental effects: Adv. Chem. Ser. 120, entitled “Chlorodioxins:
Origin and Fate,” E. H. Blair, Ed. (ACS, Washington DC, 1973) 141
pp. Toxicity and metabolism: B. A. Schwetz et al., ibid. 55-69; A. Poland, A. Kende, Fed.
Proc. 35, 2404 (1976). Special issue: Chem. Eng. News 61 (June 6, 1983).
Review of toxicology and human exposure: Toxicological Profile for 2,3,7,8-Tetradichlorodibenzo-p-dioxin (PB89-214522, 1989) 135 pp; of epidemiological data: L. Tollefson, Regul. Toxicol. Pharmacol. 13, 150-169 (1991); of receptor binding and mechanism of toxicity:
J. P. Whitlock, Jr., Annu. Rev. Pharmacol.
Toxicol. 30, 251-277 (1990); of carcinogenicity:
J. Huff et al., ibid. 34, 343-372
(1994). Review of GC high resolution mass spectrometric analysis:
J.-F. Focant et al., J. Chromatogr.
A 1067, 265-275 (2005).
Properties
Needles, mp 295° (Tomita); crystals from anisole, mp 320-325° (Sandermann). LD50 in male, female rats (mg/kg): 0.022, 0.045 orally (Schwetz).Note
An industrial accident during the manufacture
of 2,4,5-trichlorophenol in Seveso, Italy on July 10, 1976 caused
the release of an estimated two to ten pounds of TCDD into the environment.
Concentrations as high as 51.3 ppm TCDD were found in some samples:
R. Rawls, D. A. O'Sullivan, Chem. Eng. News 54, 27 (Aug. 23, 1976); A. Hay, Nature 262, 636 (1976). TCDD, as a contaminant
created in the manufacture of Agent Orange, a widely used defoliant in Vietnam during the 1960's,
has also been implicated as the causative agent of various symptoms
described by veterans exposed to the defoliant, see C.
Holden, Science 205, 770 (1979).Caution
Potential symptoms of overexposure
to TCDD are eye irritation; allergic dermatitis, chloracne; porphyria;
GI disturbances; possible reproductive and teratogenic effects. See NIOSH Pocket Guide to Chemical
Hazards (DHHS/NIOSH 97-140, 1997) p 298. Toxic effects
in animals include the wasting syndrome, gastric ulcers, immunotoxicity,
hepatotoxicity, hepatoporphyria, vascular lesions, chloracne, teratogenicity,
fetotoxicity, impaired reproductive performance, endometriosis and
delayed death. See Poland, Kende, loc. cit.; C. D. Carter et al., Science 188, 738 (1975). This substance is listed as a known
human carcinogen: Report on Carcinogens,
Eleventh Edition (PB2005-104914, 2004) p III-241.