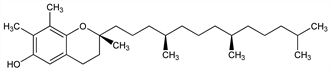
9497. γ-Tocopherol
Nomenclature
Description and references
One of the naturally occurring forms of vitamin E, q.v. Most abundant tocopherol in soybean and corn oils. Isoln by fractional crystn: Emerson et al., Science 83, 421 (1936); J. Biol. Chem. 113, 319 (1936); J. G. Baxter et al., J. Am. Chem. Soc. 65, 918 (1943). Prepn of crystalline natural form: C. D. Robeson, J. Am. Chem. Soc. 65, 1660 (1943). Comparison of bioactivity with α-tocopherol, q.v.: J. G. Bieri, R. P. Evarts, J. Nutr. 104, 850 (1974). Protective effects vs reactive nitrogen oxide species: R. V. Cooney et al., Proc. Natl. Acad. Sci. USA 90, 1771 (1993); S. Christen et al., ibid. 94, 3217 (1997). HPLC determn in serum: A. Sobczak et al., J. Chromatogr. B 730, 265 (1999). Review of bioavailability, metabolism, and activity: Q. Jiang et al., Am. J. Clin. Nutr. 74, 714-722 (2001).