Tranexamic Acid Tablets
Action and use
Antifibrinolytic.
Definition
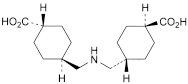
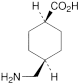
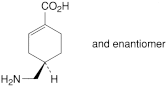
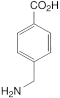
Tranexamic Acid Tablets contain Tranexamic Acid.
Content of tranexamic acid, C8H15NO2
95.0 to 105.0% of the stated amount.
Identification
Shake a quantity of the powdered tablets containing 0.5 g of Tranexamic Acid with 5 mL of water for 15 minutes, filter and add 2 mL of ether to the filtrate. Stir, add 10 mL of methanol, stir again and allow to crystallise. The crystals, after drying, comply with the following tests.
Tests
Related substances
Carry out the method for liquid chromatography, Appendix III D, using the following solutions.
Inject solution (1) and allow the chromatography to proceed for 3 times the retention time of tranexamic acid (about 13 minutes). Identify the impurities from the chromatogram obtained with solution (4) and from the reference chromatogram supplied with tranexamic acid impurity standard BPCRS.
Dissolve 11.0 g of anhydrous sodium dihydrogen orthophosphate in 500 mL of water, add 5 mL of triethylamine and 1.4 g of sodium dodecyl sulfate, adjust the pH to 2.5 with 2m orthophosphoric acid and add sufficient water to produce 600 mL. Add 400 mL of methanol and mix.
The test is not valid unless:
the chromatogram obtained with solution (4) closely resembles the reference chromatogram supplied with tranexamic acid impurity standard BPCRS;
the resolution factor between the peaks corresponding to tranexamic acid and impurity C in the chromatogram obtained with solution (4) is at least 2.0.
In the chromatogram obtained with solution (1):
the area of any peak corresponding to impurity A is not greater than the area of the principal peak in the chromatogram obtained with solution (2) (1%);
the area of any peak corresponding to impurity B is not greater than 0.5 times the area of the principal peak in the chromatogram obtained with solution (2) (0.5%);
the area of any peak corresponding to impurity C is not greater than the area of the principal peak in the chromatogram obtained with solution (3) (0.1%);
the area of any peak corresponding to impurity D is not greater than the area of the principal peak in the chromatogram obtained with solution (3) (0.1%);
the area of any other secondary peak is not greater than 0.1 times the area of the principal peak in the chromatogram obtained with solution (2) (0.1%).
Assay
Weigh and powder 20 tablets. Dissolve a quantity of the powdered tablets containing 0.25 g of Tranexamic Acid in 50 mL of anhydrous acetic acid and carry out Method I for non-aqueous titration, Appendix VIII A, using crystal violet solution as indicator. Each mL of 0.1m perchloric acid VS is equivalent to 15.72 mg of C8H15NO2.