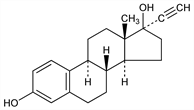
3734. Ethinyl Estradiol
Nomenclature
Description and references
Synthetic steroid with high oral estrogenic potency: Inhoffen, Hohlweg, Naturwissenschaften 26, 96 (1938). Prepn from estrone: Inhoffen et al., Ber. 71, 1024 (1938). See also DE 702063; GB 516444; US 2243887; US 2251939; US 2265976; US 2267257. Properties: Petit, Muller, Bull. Soc. Chim. Fr. 1951, 121; L. Ehmann, A. Wettstein, Pharm. Acta Helv. 25, 297 (1950). NMR: Hampel, Kraemer, Ber. 98, 3255 (1965). Toxicity: E. I. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971). Randomized double-blind clinical studies: S. Koetsawang et al., Contraception 25, 231 (1982); A. Sheth et al., ibid. 243. Clinical evaluation in gonadal dysgenesis: L. Cuttler et al., J. Clin. Endocrinol. Metab. 60, 1087 (1985). General review: K. W. Thompson, J. Clin. Pharmacol. 8, 1088-1098 (1948). Review of metabolism and pharmacokinetics: K. Fotherby, Methods Find. Exp. Clin. Pharmacol. 4, 133-141 (1982); of carcinogenicity studies: IARC Monographs 21, 233-255 (1979); ibid. Suppl. 4, 186-188 (1982).
Derivative
Hemihydrate.Properties
Fine needles from methanol + water, mp 141-146°, [α]D25 0 ± 1° (dioxane). Dehydrates after melting and further heating, mp 182-184°. [α]D24 +3.5 ± 0.5° (c = 2 in dioxane); -29.5 ± 1° (c = 2 in pyridine). uv max (ethanol): 281 nm (ε 2040 ± 60). Practically insol in water. Soly: 1 part in 6 of ethanol, 1 in 4 of ether, 1 in 5 of acetone, 1 in 4 of dioxane, and 1 in 20 of chloroform. Sol in vegetable oils, and in solns of fixed alkali hydroxides. LD50 in rats, mice (mg/kg): 2952, 1737 orally (Goldenthal).Derivative
3-Acetate.Properties
Crystals, mp 152-153°. [α]D20 +3° (chloroform).Derivative
3-Benzoate.Properties
Needles from methanol, mp 200-202°.Note
Also used in combination with chlormadinone acetate, desogestrel, ethynodiol, gestodene, lynestrenol, norethindrone or norgestrel, q.q.v. Has been used in combination with dimethisterone, medroxyprogesterone, or megestrol acetate, q.q.v.Caution
These substances are listed as known human carcinogens: Report on Carcinogens, Eleventh Edition (PB2005-104914, 2004) p III-115.Therapeutic Category
Estrogen. In combination with progestogen as oral contraceptive.
Therapeutic Category (Veterinary)
Estrogen.Keywords
Contraceptive (Oral); Estrogen; Steroidal